Made in Germany.



One unified, AI-powered procurement platform built for the complexity of regulated chemical and pharmaceutical supply chains





Go-live in under 3 weeks
<85% Tier-1 suppliers onboarded within 2 weeks


“We saw Mercanis as a platform we could grow with. It gave us what we needed right away, while still offering a vision for more automation and AI in the future.”

Go-live in under 3 weeks
<85% Tier-1 suppliers onboarded within 2 weeks


“We saw Mercanis as a platform we could grow with. It gave us what we needed right away, while still offering a vision for more automation and AI in the future.”
Mercanis standardizes GMP/GxP, REACH, CLP and SDS requirements into one guided onboarding flow. Suppliers fill out structured forms, upload required documents, and Mercanis validates formats, dates, and evidence automatically.

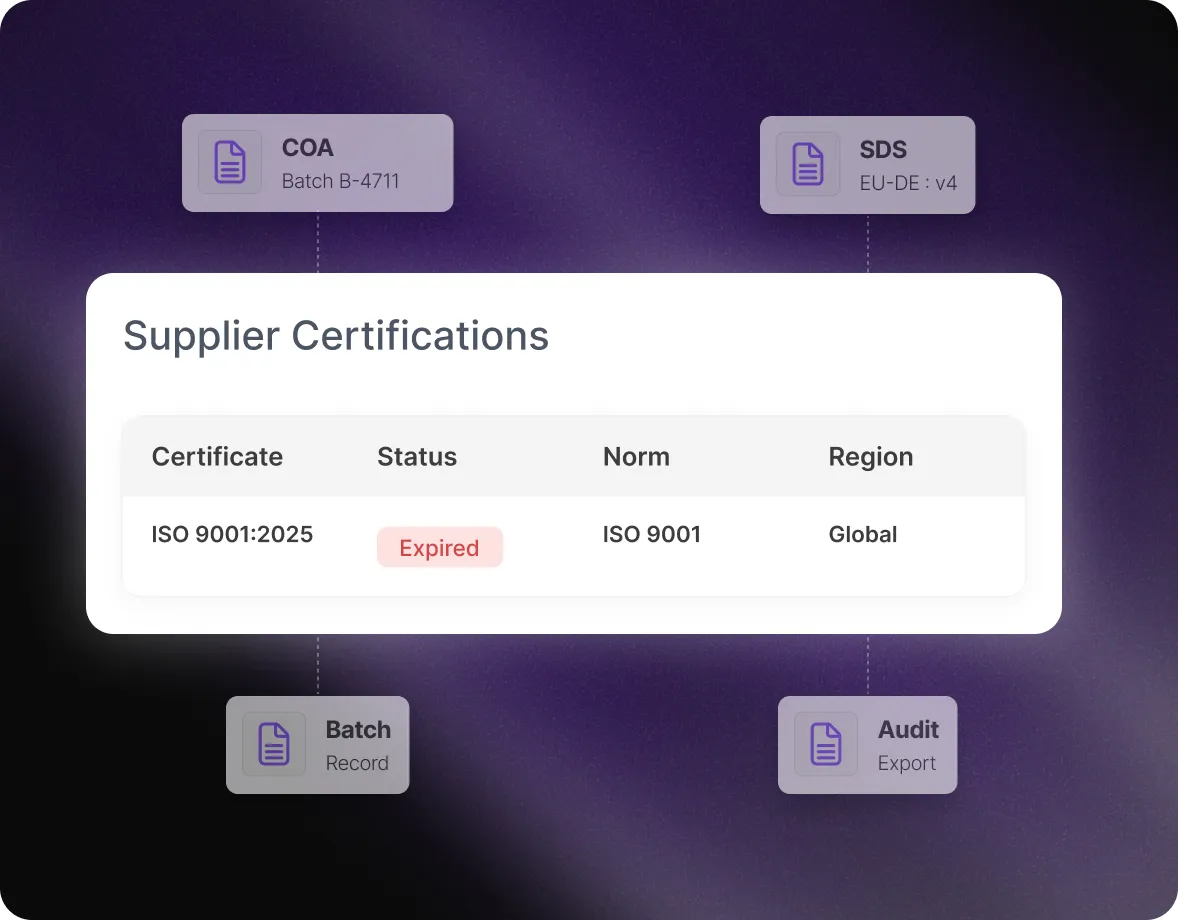
Mercanis connects every delivered batch to the correct COA, SDS version, supplier evidence, and relevant regional safety data.
QA sees a clear “release ready” status, linked documentation, and deviation alerts - enabling fast, traceable approvals.
Mercanis continuously monitors expiries, version changes, missing evidence, and documentation gaps across suppliers and batches.
Teams get early alerts - before expired SDS blocks production or outdated documents trigger an audit finding.
.webp)
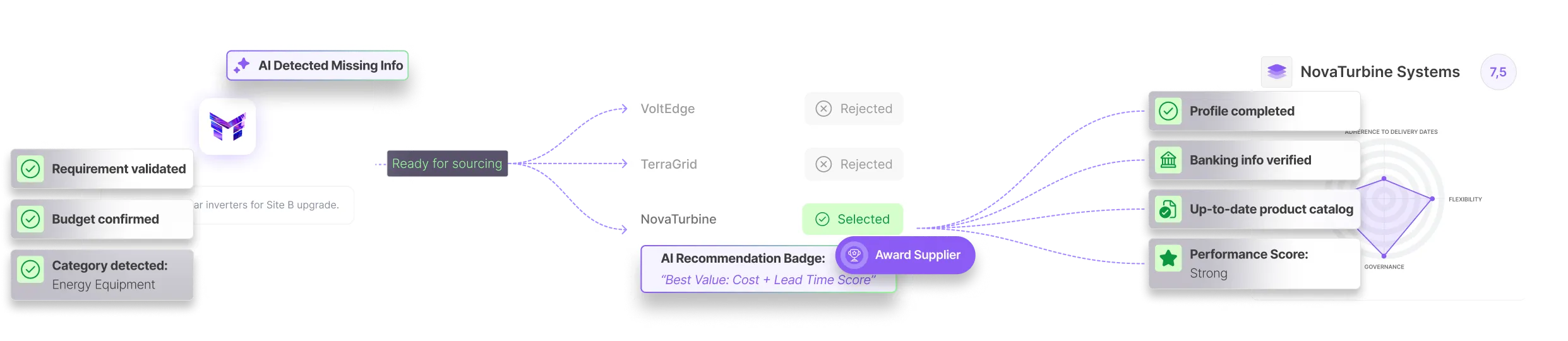
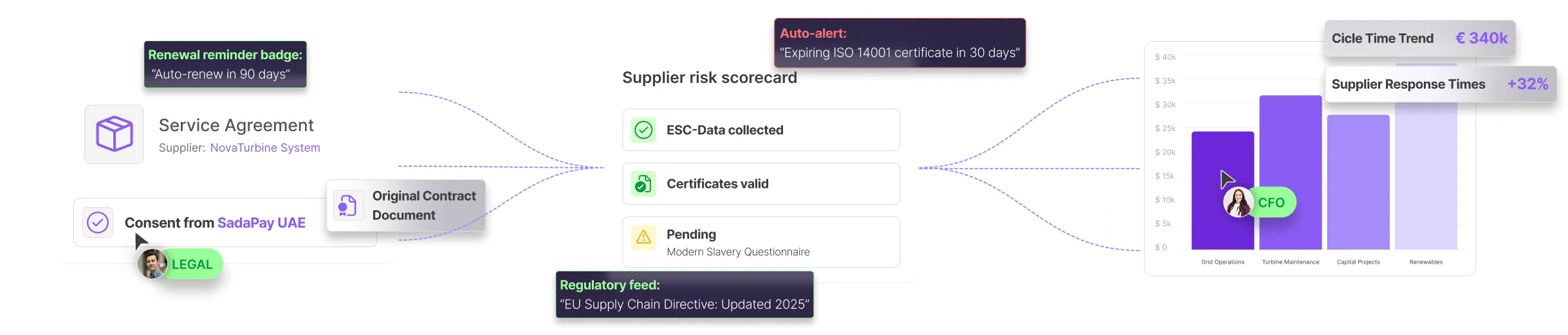




Mercanis standardizes documentation requirements, tracks expiries, and creates full audit trails for every supplier. This ensures qualification and sourcing workflows follow GMP and GxP standards.

Yes. Mercanis monitors all document versions, expiry dates and regional SDS requirements. Suppliers are reminded automatically to update missing or outdated files.

Preconfigured templates collect GMP, REACH, CLP, ISO and safety data in one structured flow. QA, Regulatory and Procurement share the same validated supplier record.

Absolutely. Mercanis provides complete evidence packages, version histories, approval logs and compliance summaries — ready for internal or external inspections.